-
 * * HOW TO DETERMINE AAS HALF LIFE - MEMBER DISCUSSION THREAD * *
* * HOW TO DETERMINE AAS HALF LIFE - MEMBER DISCUSSION THREAD * *
On the regular I get asked about the half lives of certain gear and how the half elimination period is determined.
As far as I know there is a direct correlation between the # of carbons and the half life.
To start I've provided a list of esters and how many carbons are in the carbon chain. If you have different data or this list is outdated then pls let me know.
Here's a brief explanation on the knowledge I have to date.
Steroid esters are a steroid molecule with a carbon chain attached. Each ester is unique having its own # of carbon atoms.
The carbon chain, is directly involved in determining the half-life of the steroid, and solubility in the bloodstream.
So let's look at the list that I believe to be most accurate.
Formate-1 carbon ester
Acetate-2 carbon ester
Propionate-3 carbon ester
Butyrate-4 carbon ester
Valerate-5 carbon ester
Hexanoate-6 carbon ester
Heptanoate-7 carbon ester
Enanthate-7 carbon ester
Octanoate-8 carbon ester
Cypionate-8 carbon ester
Nonanoate-9 carbon ester
Decnoate-10 carbon ester
Undecanoate-11 carbon ester
The higher the # of carbon atoms in the ester, the less soluble the steroid is in the blood stream, and the longer the half-life. Two enzymes...esterase and hydrolysate...cleave off the carbon atoms from the carbon chain.
Finally the carbon atoms are removed and the steroid is activated.
HERE'S WHAT I NEED TO KNOW AND CAN'T FIGURE OUT...."IS THE # OF CARBON ATOMS IN THE CHAIN THE ONLY DETERMINING FACTOR OR IS THERE MORE TO THIS?"
Pls guys offer up all you know, can find thru a search online, or asking fellow hobbyists. Post any and all you can find. It might seem insignificant only to prove a key piece of the puzzle at a later date.
This is a thought provoking thread. Meaning I have acquired some knowledge over the last year & a half on the subject of pharmacokinetics for the purpose of knowing how to determine the half/full elimination periods of the AAS we commonly use. If you think I'm wrong or misinformed then I encourage you to show me the inaccuracies or identify the data, I have provided, that is outdated. What would be ideal in that situation is to simultaneously educate me on this subject and tell me exactly how to determine the elimination period so I can do it on my own ......aaaaand my search will end!
Point is no judgment in this thread or drama. This is for the purpose of seeking knowledge and sharing knowledge. Whatever you've got or can find pls POST IT.
If an answer is not posted soon then who knows maybe in a few yrs a member will have an answer for us.
Now let's compile some info......
Last edited by Riggs; 09-08-2020 at 07:33 AM.
-
Post Thanks / Like - 7 Thanks, 7 Likes, 0 Dislikes
----
-
Just some old stuff I came across looking through my notes:
ARTICLES CURRENTLY RESEARCHING
Half-life explicitly describes the time it takes for the blood plasma concentration of a substance to halve (plasma half-life) its steady-state when circulating in the full blood of an organism. This measurement is useful in medicine and pharmacology because it helps determine how much of a drug needs to be taken and how frequently it needs to be taken if a certain average amount is needed constantly. In contrast, the stability of a substance direct in plasma is described with plasma stability that is essential to ensure accurate analysis of drugs in plasma and for Drug discovery.
The relationship between the biological and plasma half-lives of a substance can be complex depending on the substance in question, due to factors including accumulation in tissues (protein binding), active metabolites, and receptor interactions. The amount taken, route of administration, preparation , metabolism, environment. So all the charts listed are guesses.
Just a note about Absorption of Esters; below is a study of Enanthate vs. Cypionate as studied by the NIH scientists:
The pharmacokinetic profile of testosterone enanthate was studied in a regime of multiple dosing and the testosterone level was reported to present a Cmax above 1200 ng/dl after 24 hours of the last dose. The concentration decreased sequentially until it reached 600 ng/dl after one week. The pharmacokinetic profile of testosterone enanthate presented differences depending on the administered dose in which the tmax was shifted to a range of 36-48 hours. The plasma testosterone level plateaued below the therapeutic range after 3-4 weeks. This reports showed that the different formulation of testosterone enanthate and testosterone cypionate generates a different profile and thus, they are not therapeutically equivalent.
Ester info: Steroid esters are compounds containing a steroid moiety which bears a carboxylic acid ester group.
Testosterone propionate 4.5 days
Testosterone enanthate presents a long half-life in the range of 7-9 days
Testosterone cypionate: The half-life of testosterone cypionate is one of the longest, being approximately of 8 days
It was developed by the company Pharmacia and Upjohn
*Testosterone Acetate: could not be determined due to lack of scientific data (although I saw roid sites saying longer than propionate)*The drug was first described in 1936 and was one of the first androgen esters and esters of testosterone to be synthesized. It is hydrophobic; practically insoluble in water
Testosterone no ester: elimination half life: 2-4hrs.
This is just some stuff. So many factors and things happen inside our bodies and there are tiny, little differences with compounds that change inside of plus or in the lab by adding or not adding a certain part of the science.
Max
-conflicting/confusing usually comes down to money w studies but science is science.....or is it??
-
Post Thanks / Like - 5 Thanks, 5 Likes, 0 Dislikes
----
-
I wonder if any of the boys who brew or who understand the process of manufacturing raws will have some interesting information.
-
Post Thanks / Like - 3 Thanks, 3 Likes, 0 Dislikes
----
-

Originally Posted by
Mattymoo

I wonder if any of the boys who brew or who understand the process of manufacturing raws will have some interesting information.
Right? But they are in another subforum. It all has play action though. I am fascinated! 

Max
-
Post Thanks / Like - 1 Thanks, 2 Likes, 0 Dislikes
----
-

Originally Posted by
Mattymoo

I wonder if any of the boys who brew or who understand the process of manufacturing raws will have some interesting information.
Not one that I've worked with or talked to knows anything about Pharmacokinetics.
-
Post Thanks / Like - 2 Thanks, 2 Likes, 0 Dislikes
----
-

Originally Posted by
maxmuscle1

Just some old stuff I came across looking through my notes:
ARTICLES CURRENTLY RESEARCHING
Half-life explicitly describes the time it takes for the blood plasma concentration of a substance to halve (plasma half-life) its steady-state when circulating in the full blood of an organism. This measurement is useful in medicine and pharmacology because it helps determine how much of a drug needs to be taken and how frequently it needs to be taken if a certain average amount is needed constantly. In contrast, the stability of a substance direct in plasma is described with plasma stability that is essential to ensure accurate analysis of drugs in plasma and for Drug discovery.
The relationship between the biological and plasma half-lives of a substance can be complex depending on the substance in question, due to factors including accumulation in tissues (protein binding), active metabolites, and receptor interactions. The amount taken, route of administration, preparation , metabolism, environment. So all the charts listed are guesses.
Just a note about Absorption of Esters; below is a study of Enanthate vs. Cypionate as studied by the NIH scientists:
The pharmacokinetic profile of testosterone enanthate was studied in a regime of multiple dosing and the testosterone level was reported to present a Cmax above 1200 ng/dl after 24 hours of the last dose. The concentration decreased sequentially until it reached 600 ng/dl after one week. The pharmacokinetic profile of testosterone enanthate presented differences depending on the administered dose in which the tmax was shifted to a range of 36-48 hours. The plasma testosterone level plateaued below the therapeutic range after 3-4 weeks. This reports showed that the different formulation of testosterone enanthate and testosterone cypionate generates a different profile and thus, they are not therapeutically equivalent.
Ester info: Steroid esters are compounds containing a steroid moiety which bears a carboxylic acid ester group.
Testosterone propionate 4.5 days
Testosterone enanthate presents a long half-life in the range of 7-9 days
Testosterone cypionate: The half-life of testosterone cypionate is one of the longest, being approximately of 8 days
It was developed by the company Pharmacia and Upjohn
*Testosterone Acetate: could not be determined due to lack of scientific data (although I saw roid sites saying longer than propionate)*The drug was first described in 1936 and was one of the first androgen esters and esters of testosterone to be synthesized. It is hydrophobic; practically insoluble in water
Testosterone no ester: elimination half life: 2-4hrs.
This is just some stuff. So many factors and things happen inside our bodies and there are tiny, little differences with compounds that change inside of plus or in the lab by adding or not adding a certain part of the science.
Max
-conflicting/confusing usually comes down to money w studies but science is science.....or is it??
What you've summarized or quoted states Test P half life at or around 4.5 days. So does Pubchem which I love on NCBI.
Screenshot_20200908-064035_Samsung Internet Beta.jpg
Wikipedia states Test P half life is .8 days or 19 hours.
Screenshot_20200908-063711_Samsung Internet Beta.jpg
Which is it?
Starting to see why I said "fuck this I'm gonna find out how to determine the elimination periods on my own.
Last edited by Riggs; 09-08-2020 at 11:44 AM.
-
Post Thanks / Like - 1 Thanks, 1 Likes, 0 Dislikes
----
-

Originally Posted by
maxmuscle1

The pharmacokinetic profile of testosterone enanthate was studied in a regime of multiple dosing and the testosterone level was reported to present a Cmax above 1200 ng/dl after 24 hours of the last dose. The concentration decreased sequentially until it reached 600 ng/dl after one week.
I read the same study in an Upjohn Test C leaflet.
This is the exact study I read that prompted me to pin high carbon chain esters more frequently. I had always said that even with long esters there's an initial spike in blood plasma levels so when I read this I finally had something to back my claim.
As a matter of fact I found the leaflet and mentioned the results of the study to Weapon while discussing the topic of this thread maybe a yr ago.
Last edited by Riggs; 09-08-2020 at 11:33 AM.
-
Post Thanks / Like - 1 Thanks, 1 Likes, 0 Dislikes
----
-
-
Post Thanks / Like - 1 Thanks, 1 Likes, 0 Dislikes
 Riggs
Riggs thanked for this post
----
-
----
-

Originally Posted by
maxmuscle1

I do believe that both screenshots I provided are in reference to Biological Half Life but I'm honestly not sure now. I need to go back and look again.
-
Post Thanks / Like - 1 Thanks, 1 Likes, 0 Dislikes
----




















































 Reply With Quote
Reply With Quote







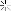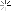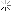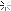
Bookmarks